Distillation separates a fluid containing two or more components into its individual parts. Common examples are refining crude oil into its many elemental substances and separating alcohol from fermented liquid to make alcoholic beverages. Alcohol boils at 172°F, water at 212°F. A mixture of alcohol and water boils somewhere between these temperatures depending on the amount of each the solution contains. Vapor given off from boiling contains both alcohol and water, more alcohol than water at first because of its lower boiling point, but an ever-declining amount as the boiling point rises due to diminishing alcohol in the mixture. Multiple distillations thus achieve higher and higher percentages of alcohol because the fluid contains less water each time. Two common distillation processes are used for alcoholic beverages.
POT STILL
 A commercial pot still is an onion- or flask-shaped vessel wide at the bottom, narrowing toward the top, with an outward-pointing beak. Is is usually made of copper which maintains an even temperature during heating. A mixture such as fermented grain mash (the “wash”) is poured into the still which is then heated to boiling. Vapor rises, flows into and through the beak, then passes through a cooling chamber where it condenses and is collected in another container. This continues until the remaining wash is mostly water, whereupon the still is cooled, emptied and refilled, and the process begins again. For this reason it is called batch distillation. Condensate contains about 30% alcohol after the first distilling, along with a variety of aroma and flavor elements of the wash. Additional distillations increase the alcohol percentage and either concentrate or remove additional elements from the wash.
A commercial pot still is an onion- or flask-shaped vessel wide at the bottom, narrowing toward the top, with an outward-pointing beak. Is is usually made of copper which maintains an even temperature during heating. A mixture such as fermented grain mash (the “wash”) is poured into the still which is then heated to boiling. Vapor rises, flows into and through the beak, then passes through a cooling chamber where it condenses and is collected in another container. This continues until the remaining wash is mostly water, whereupon the still is cooled, emptied and refilled, and the process begins again. For this reason it is called batch distillation. Condensate contains about 30% alcohol after the first distilling, along with a variety of aroma and flavor elements of the wash. Additional distillations increase the alcohol percentage and either concentrate or remove additional elements from the wash.
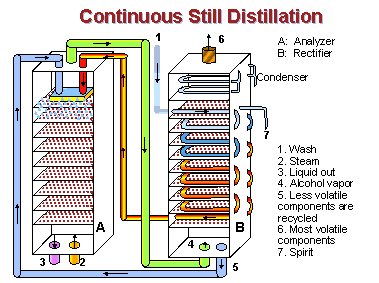
The column still was invented in 1826 by a Scottish distiller, then improved and patented five years later by the Irish inventor and distiller, Aeneas Coffey. Known also as the Coffey or continuous still, it consists of two columns: the “analyzer” and the “rectifier.”
The analyzer contains a series of perforated plates stacked about a foot apart. As heated wash flows into the top of the column and drips down through the perforated plates, steam forced in at the column’s base rises through the plates from below. As the two meet on the plates, the wash boils and a mixture of alcohol vapors and steam rises to the top of the column. Wash containing an ever-diminishing amount of alcohol continues down through the plates and drains off at the bottom.
The rectifier column also contains perforated plates but stacked closer together. Hot vapors from the analyzer enter at the rectifier’s base and rise through the plates which support a cooling coil spiraling down from the top. This accomplishes two things: 1) vapor begins to condense on the cooling coil, and 2) vapor heats the cooling coil through which a continuous stream of new cold wash is flowing on its way to the analyzer.
The most volatile and undesirable alcohol vapors vent away through the rectifier’s top. The remaining vapor condenses into alcohol at the top and flows into another container. The least volatile components, such as water, drip down from the coil through the plates and are returned to the analyzer. Most flavor and aroma elements from the wash are removed by this process so the alcohol is virtually tasteless. Column stills in modern distilleries are 40 feet tall or more and will produce alcohol continuously as long as new wash flows into the still.
_______________
Image of continuous still from stillcooker.com.